MARTENSITE
Martensite, named after the German metallurgist Adolf Martens (1850–1914), is any crystal structure that is formed by displacive transformation, as opposed to much slower diffusive transformations. It includes a class of hard minerals occurring as lath- or plate-shaped crystal grains. When viewed in cross-section, the lenticular (lens-shaped) crystal grains appear acicular (needle-shaped), which is how they are sometimes incorrectly described. “Martensite” most commonly refers to a very hard constituent of steel (the alloy of iron and carbon) important in some tool steels. The martensite is formed by rapid cooling (quenching) of austenite which traps carbon atoms that do not have time to diffuse out of the crystal structure.
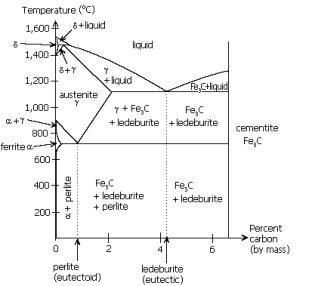
Martensite is not shown in the equilibrium phase diagram of the iron-carbon system because it is a metastable phase, the kinetic product of rapid cooling of steel containing sufficient carbon. Since chemical processes (the attainment of equilibrium) accelerate at higher temperature, martensite is easily destroyed by the application of heat. This process is called tempering. In some alloys, the effect is reduced by adding elements such as tungsten that interfere with cementite nucleation, but, more often than not, the phenomenon is exploited instead. Since quenching can be difficult to control, many steels are quenched to produce an overabundance of martensite, then tempered to gradually reduce its concentration until the right structure for the intended application is achieved. Too much martensite leaves steel brittle, too little leaves it soft.
PEARLITE
Pearlite is a two-phased, lamellar (or layered) structure composed of alternating layers of alpha-ferritecementite (12%) that occurs in some steels and cast irons. It forms by a eutectoid reaction as austenite is slowly cooled below 727°C. The eutectoid composition of Austenite is approximately 0.8% carbon [1]; steel with less carbon content will contain a corresponding proportion of relatively pure ferrite crystallites that do not participate in the eutectoid reaction and cannot transform into pearlite. (88 wt%) and
The appearance of pearlite under the microscope resembles mother of pearl (also a lamellar structure), from which it takes its name.
A similar structure with lamelle much smaller than the wavelength of visible light lacks this pearlescent appearance. Called bainite, it is prepared by more rapid cooling. Unlike pearlite, whose formation involves the diffusion of all atoms, bainite grows by a displacive transformation mechanism.
AUSTENITE
Austenite (or gamma phase iron) is a metallic non-magnetic solid solution of iron and an alloying element. In plain-carbon steel, austenite exists above the critical eutectoid temperature of 1000 K (about 727 °C); other alloys of steel have different eutectoid temperatures. It is named after Sir William Chandler Roberts-Austen (1843-1902).


BAINITE
Bainite is a mostly metallic substance that exists in steelheat treatments. First described by Davenport E. S. and Edgar Bain, it forms when austenite (a solution of carbon in iron) is rapidly cooled past a critical temperature of 723°C (about 1333°F). after certain
A fine non-lamellar structure, bainite commonly consists of ferrite and cementite. It is similar in constitution to pearlite, but with the ferrite forming by a displacive mechanism similar to martensite formation, usually followed by precipitation of carbides from the supersaturated ferrite or austenite.
When formed during continuous cooling, the cooling rate to form bainite is higher than that required to form pearlite, but lower than that to form martensite, in steel of the same composition.
(source wikipedia)















0 komentar:
Posting Komentar
Jazakallah